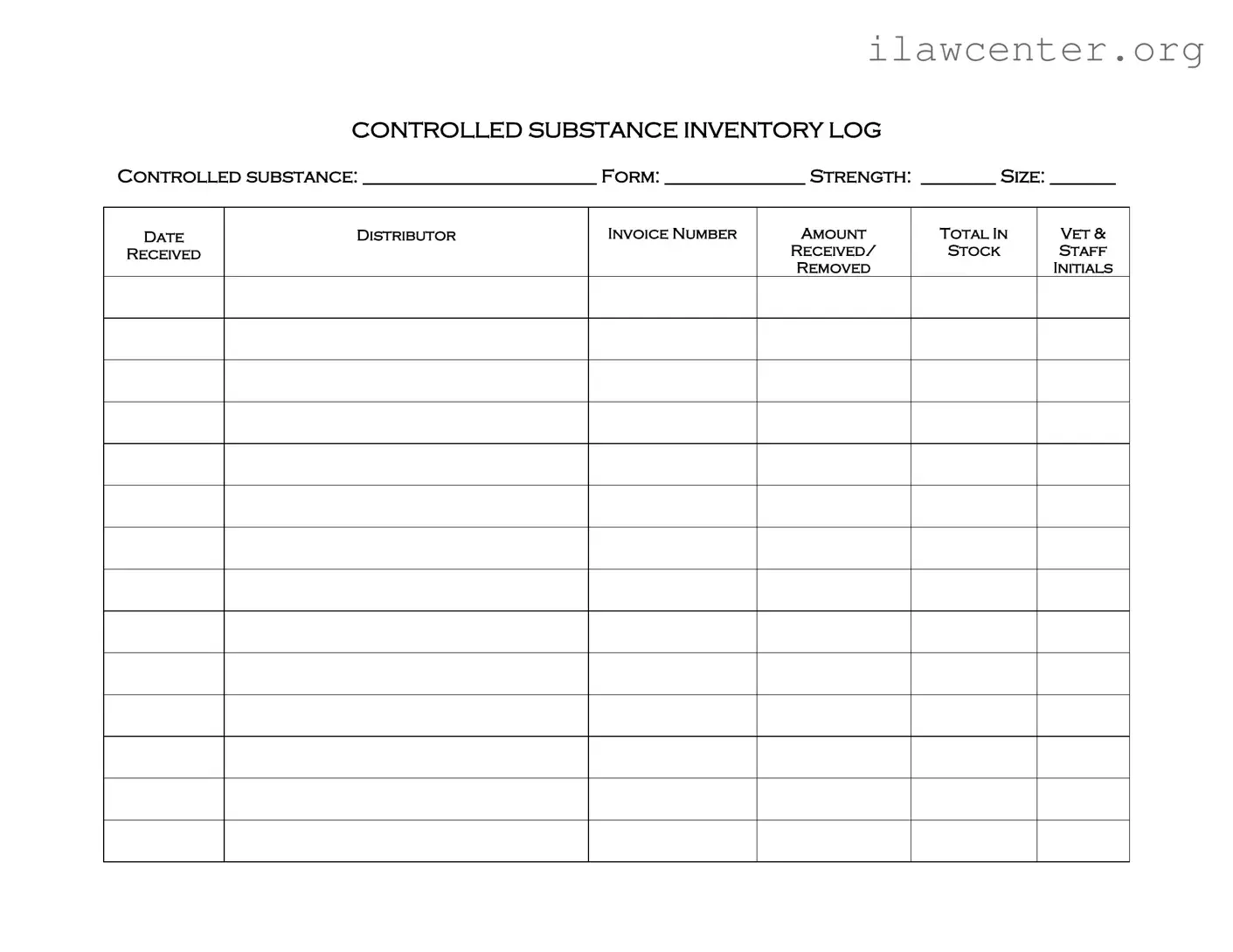
Filling out the Controlled Drug Log form can be a straightforward process, but many individuals make common mistakes that can lead to issues down the line. One frequent error is failing to complete all required fields. Each section of the form, such as controlled substance, form, strength, and size, must be filled out accurately. Omitting any information can create confusion and complicate record-keeping.
Another mistake involves incorrect data entry. For instance, entering the wrong date or invoice number can lead to discrepancies in inventory tracking. It's essential to double-check these details before finalizing the log to ensure they match the corresponding invoices and receipts.
People often overlook the importance of recording the amount received and the amount removed correctly. Miscalculating these figures can result in an inaccurate inventory count, which may raise red flags during audits. Always verify that these numbers align with physical stock levels.
Additionally, some individuals neglect to include their initials in the log after removing a controlled substance. This step is crucial for accountability and traceability. Without proper initials, it can be challenging to determine who accessed the drugs, potentially leading to liability issues.
Inconsistency in record-keeping practices is another common pitfall. Using different formats or abbreviations can create confusion. It’s best to establish a standard method for filling out the form and stick to it. Consistency helps maintain clarity and reliability in records.
Some individuals fail to update the log promptly after receiving or removing substances. Delays in recording transactions can lead to gaps in documentation. It’s advisable to fill out the log immediately to ensure that all information is current and accurate.
Another mistake is not keeping a backup of the log. Relying solely on a single copy can be risky. If the log is lost or damaged, having a backup ensures that vital information remains accessible.
People sometimes forget to train staff on how to properly complete the Controlled Drug Log form. Without proper training, errors are more likely to occur. Regular training sessions can help reinforce the importance of accuracy and compliance in documentation.
Lastly, failing to review the log regularly can lead to unnoticed mistakes. Periodic audits of the log can help catch errors early, ensuring that records remain accurate and compliant with regulations. Regular checks can prevent minor issues from escalating into significant problems.