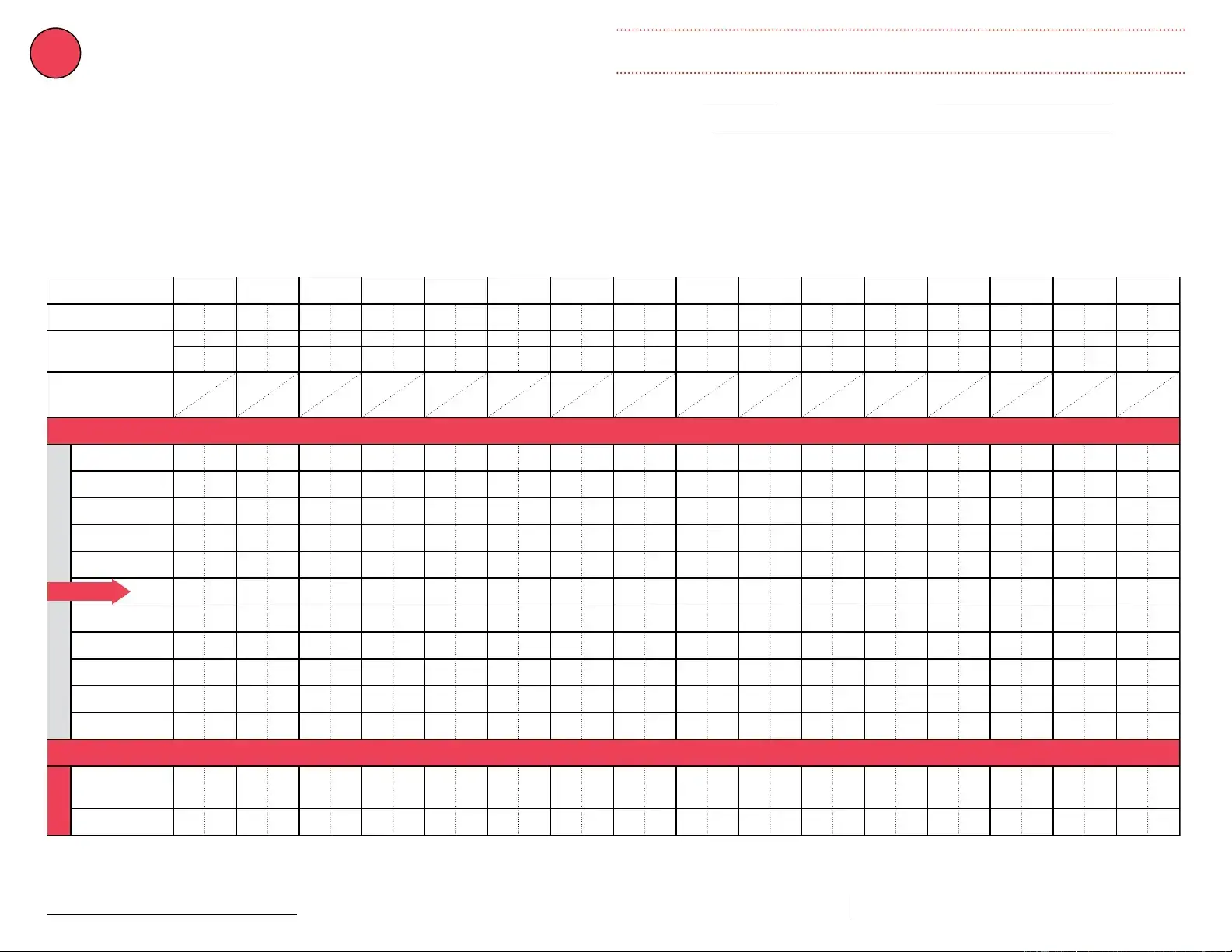
Date & Time of Event
If multiple, related events occurred,
see Description of Event below.
Storage Unit Temperature
at the time the problem was discovered
Room Temperature
at the time the problem was discovered
Person Completing Report
Date: Temp when discovered: 28º F Temp when discovered: º F Name:
Time: Minimum temp: 28º F Maximum temp: 42º F Comment (optional) Title: Date:
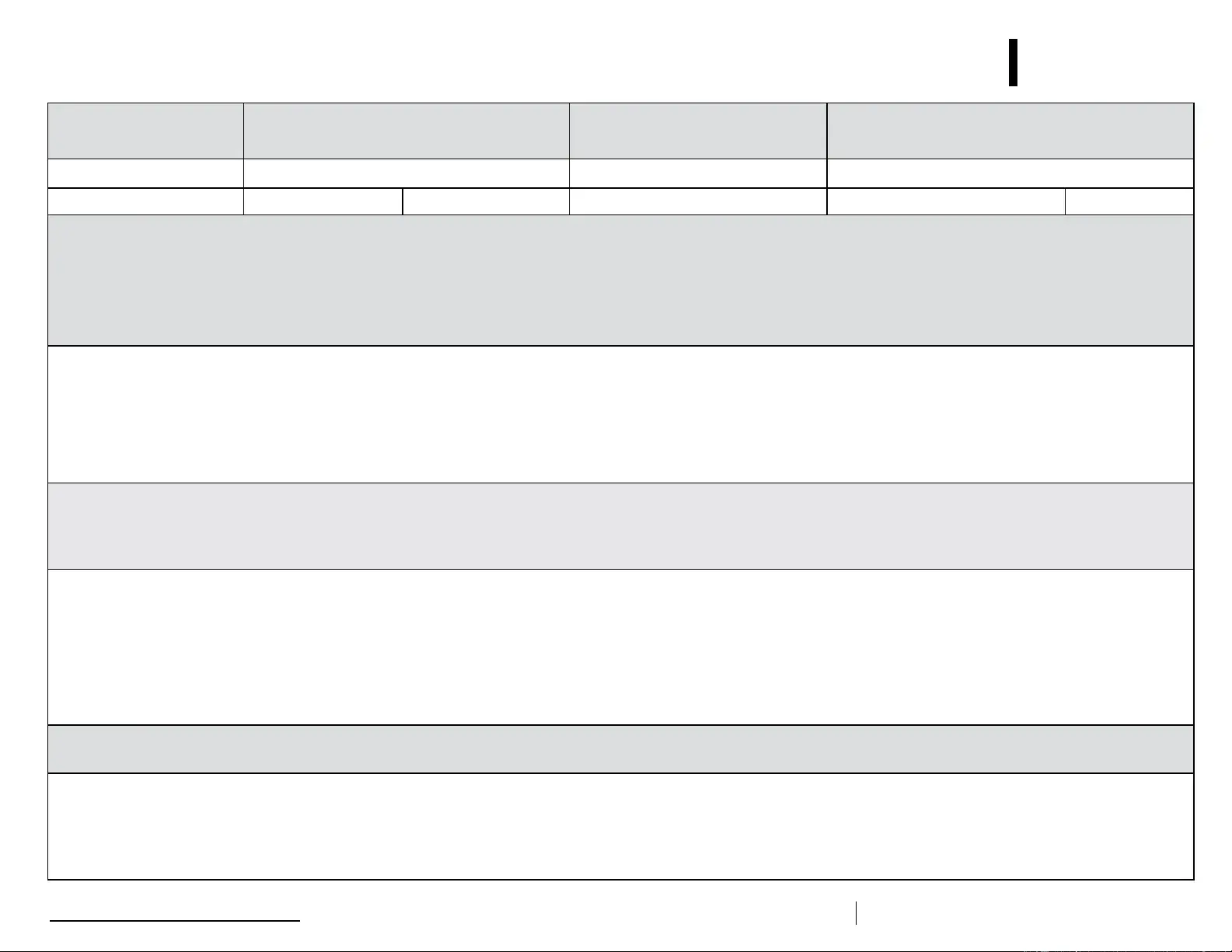
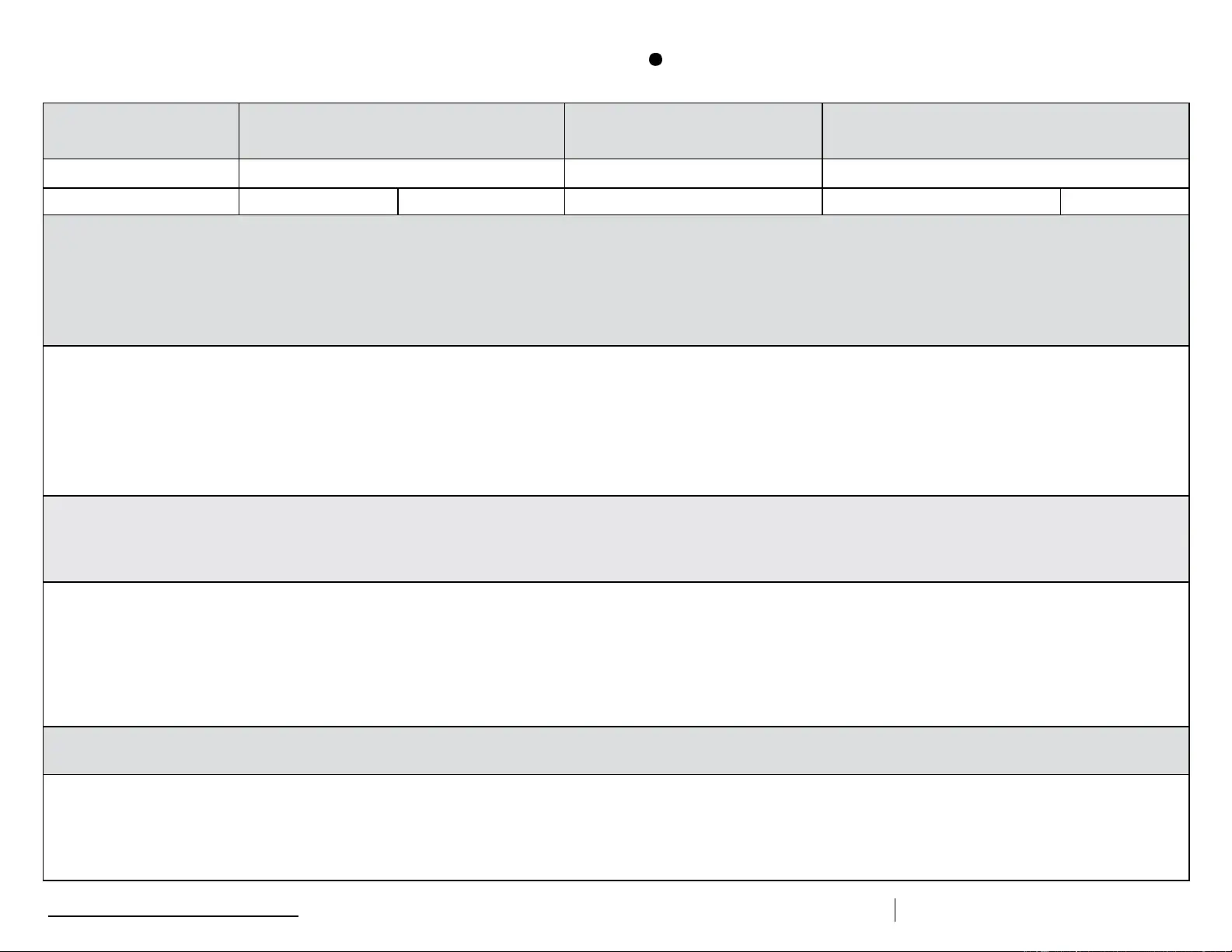
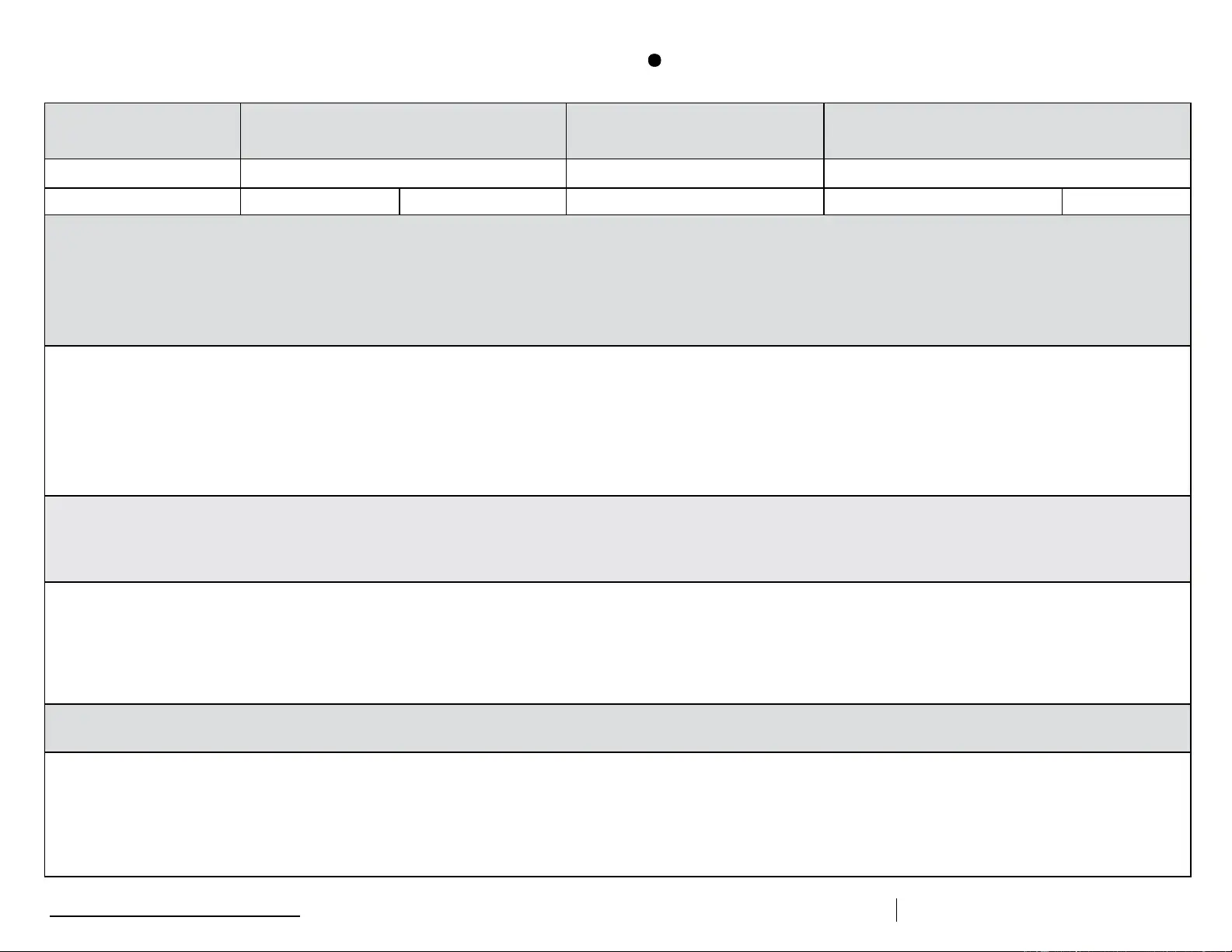
Description of Event (If multiple, related events occurred, list each date, time, and length of time out of storage.)
• General description (i.e., what happened?)
• Estimated length of time between event and last documented reading of storage temperature in acceptable range (2
o
to 8
o
C [36
o
to 46
o
F] for refrigerator; -50
o
to -15
o
C [-58º to 5ºF] for freezer; -80
o
to -60
o
C [-112º to -76ºF]
for ultra-cold freezer (Pzer COVID-19 vaccine only)
• Inventory of aected vaccines, including (1) lot #s and (2) whether purchased with public (for example, VFC) or private funds (Use separate sheet if needed, but maintain the inventory with this troubleshooting record.)
• At the time of the event, what else was in the storage unit? For example, were there water bottles in the refrigerator and/or frozen coolant packs in the freezer?
• Prior to this event, have there been any storage problems with this unit and/or with the aected vaccine?
• Include any other information you feel might be relevant to understanding the event.
Results
• What happened to the vaccine? Was it able to be used? If not, was it returned to the distributor? (Note: For public-purchase vaccine, follow your state/local health department instructions for vaccine disposition.)
ºº
º
distributed by the
Immunization Action Coalition
Saint Paul, Minnesota
•
651
-
647
-
9009
•
www.immunize.org
•
www.vaccineinformation.org
www.immunize.org/catg.d/p3041.pdf • Item#P3041(8/21)
Example
º
º
ºººF
º
Action Taken (Document thoroughly. This information is critical to determining whether the vaccine might still be viable!)
• When were the aected vaccines placed in proper storage conditions? (Note: Do not discard the vaccine. Store exposed vaccine in proper conditions and label it “do not use” until after you can discuss with your state/
local health department and/or the manufacturer[s].)
• Who was contacted regarding the incident? (For example, supervisor, state/local health department, manufacturer—list all.)
• IMPORTANT: What did you do to prevent a similar problem from occurring in the future?
Vaccine Storage Troubleshooting Record (check one)
◯
Refrigerator
◯
Freezer
◯
Ultra-Cold Freezer
Use this form to document any unacceptable vaccine storage event, such as exposure of refrigerated vaccines to temperatures that are outside the manufacturers' recommended storage ranges.